Research
Control of Tracheal Tube Size and Shape
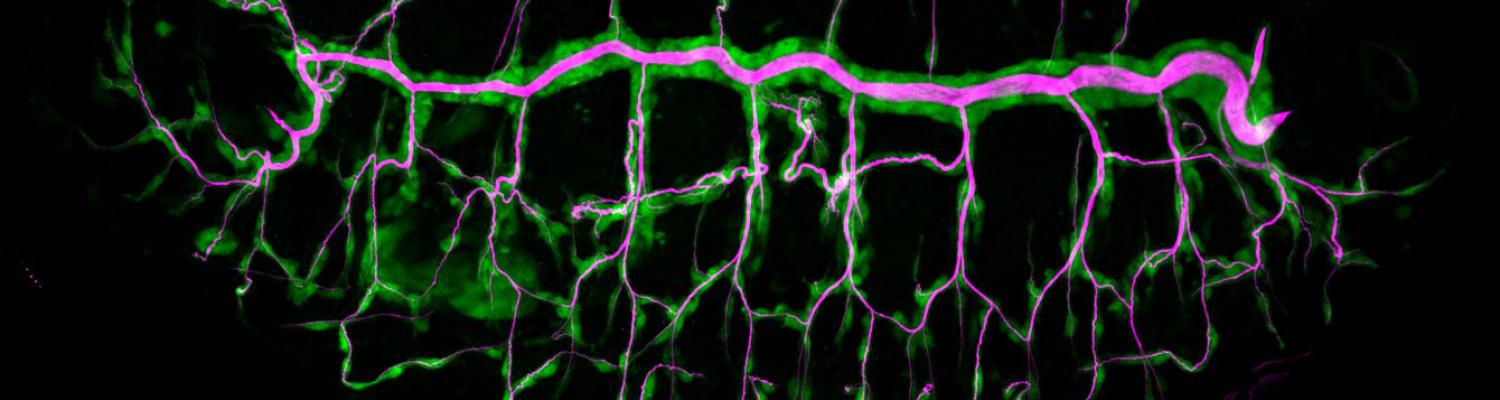
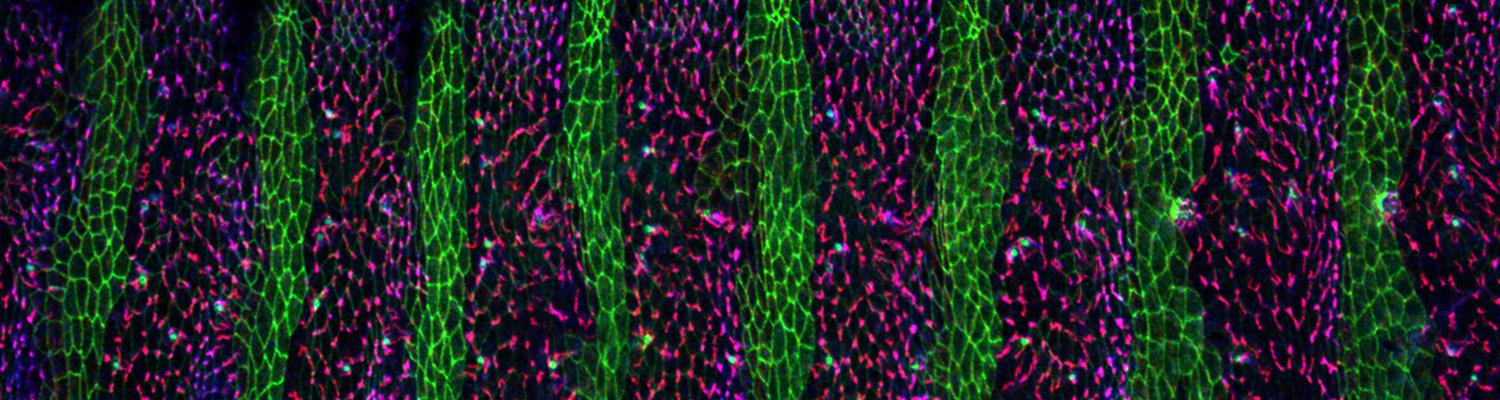
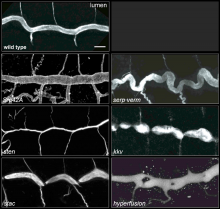
Using the tracheal system in the Drosophila embryo as a model system, we carried out systematic mutagenesis screens, in which we isolated mutations affecting various morphological features (tube length, diameter, luminal shape, tube fusion), as well as cellular processes (secretion, endocytosis, epithelial barrier function). The mutations fall into distinct phenotypic classes, thus providing an entry point towards a comprehensive analysis of the mechanisms underlying tubular morphogensis.
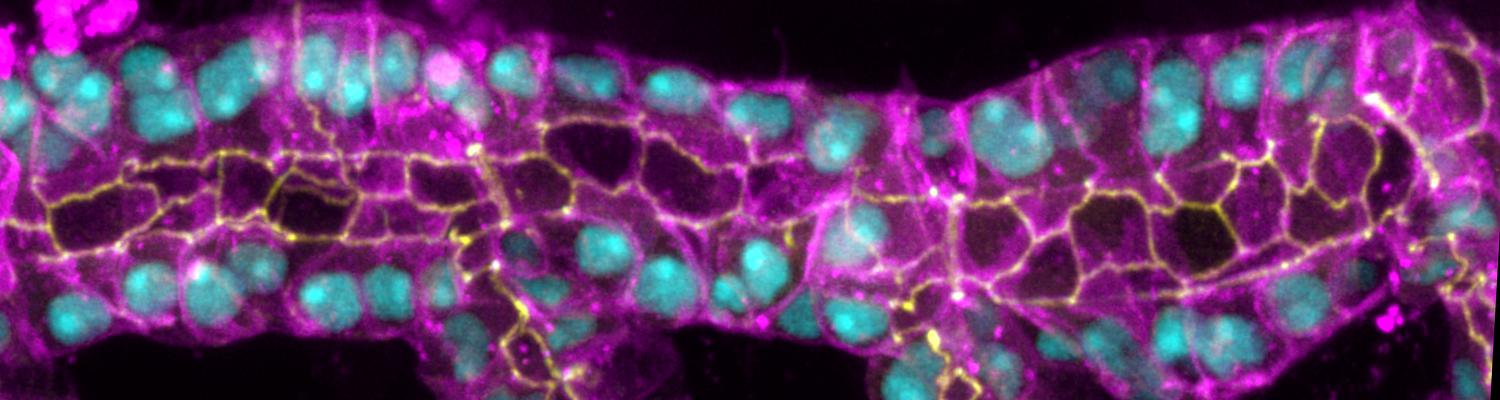
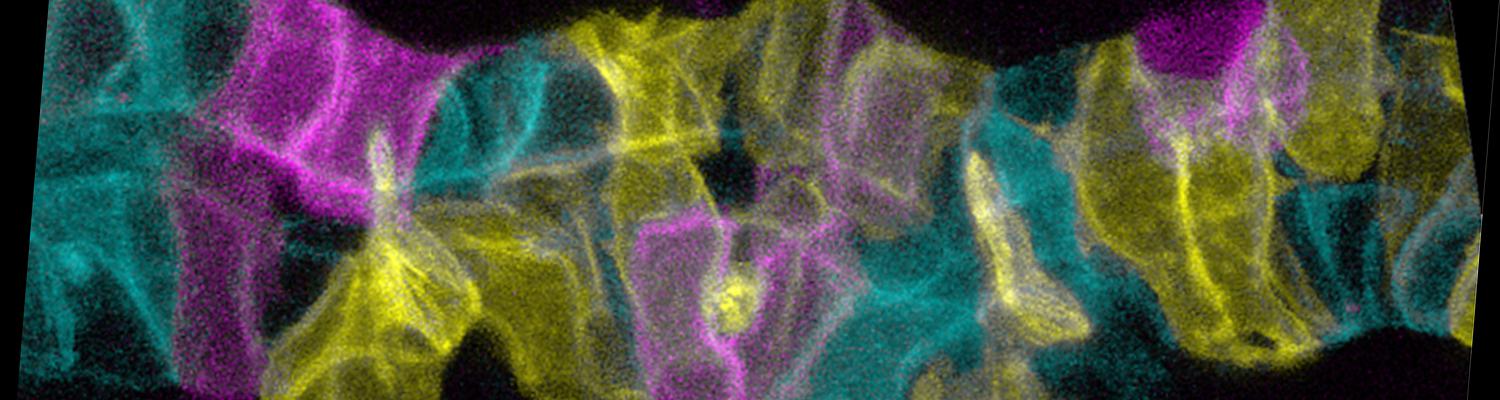
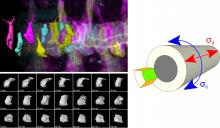
Cell Behavior and Mechanical Forces in Cylindrical Epithelia
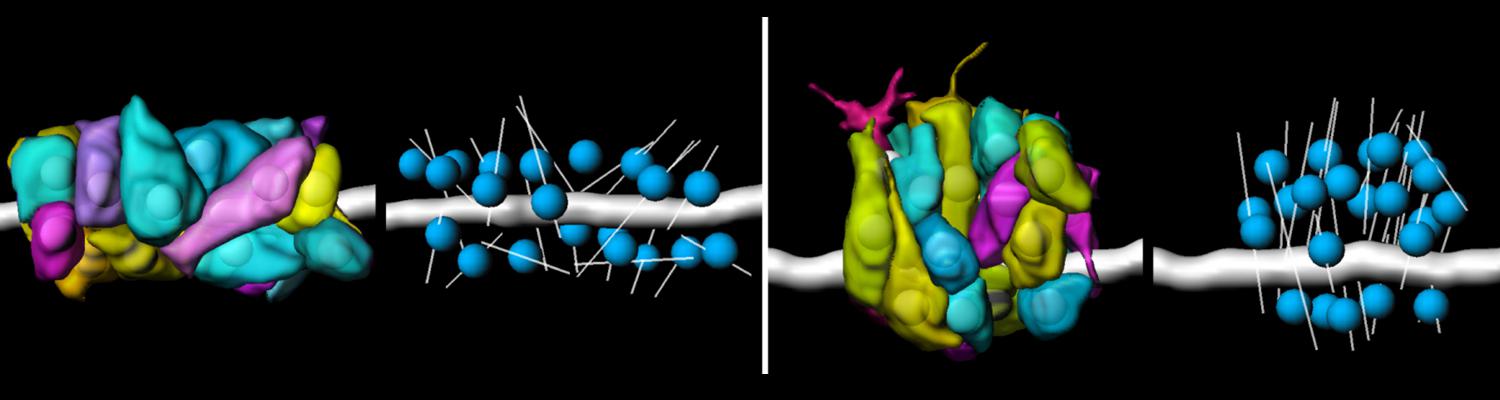
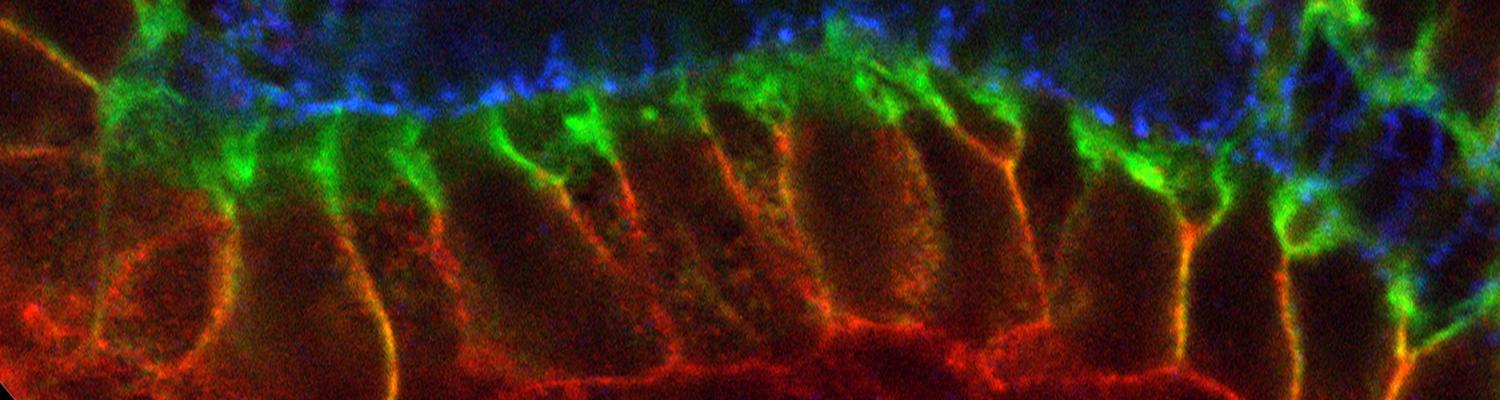
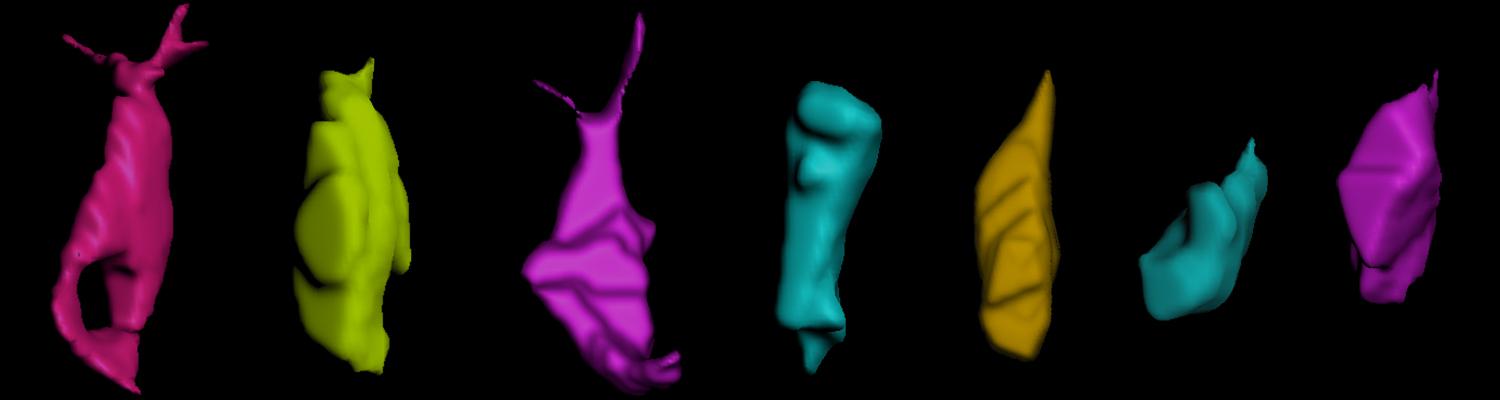
We defined distinct cellular processes that independently control tracheal tube elongation and diametric expansion. We showed that the conserved tyrosine kinase Src42 induces axially polarized cell shape changes that drive tracheal tube elongation (Förster and Luschnig 2012). Conversely, diametric tube expansion is controlled independently of Src42 by apical secretion. Secretory activity drives luminal expansion in a cell-autonomous fashion by promoting apical membrane growth and cell flattening.
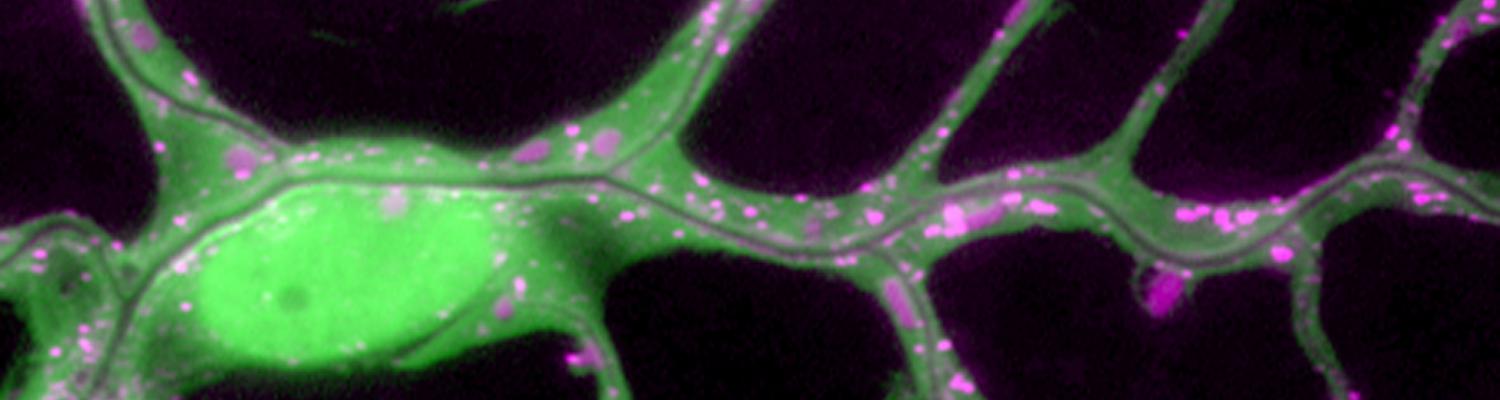
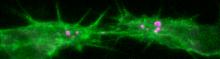
Membrane Dynamics During Epithelial Tube Fusion
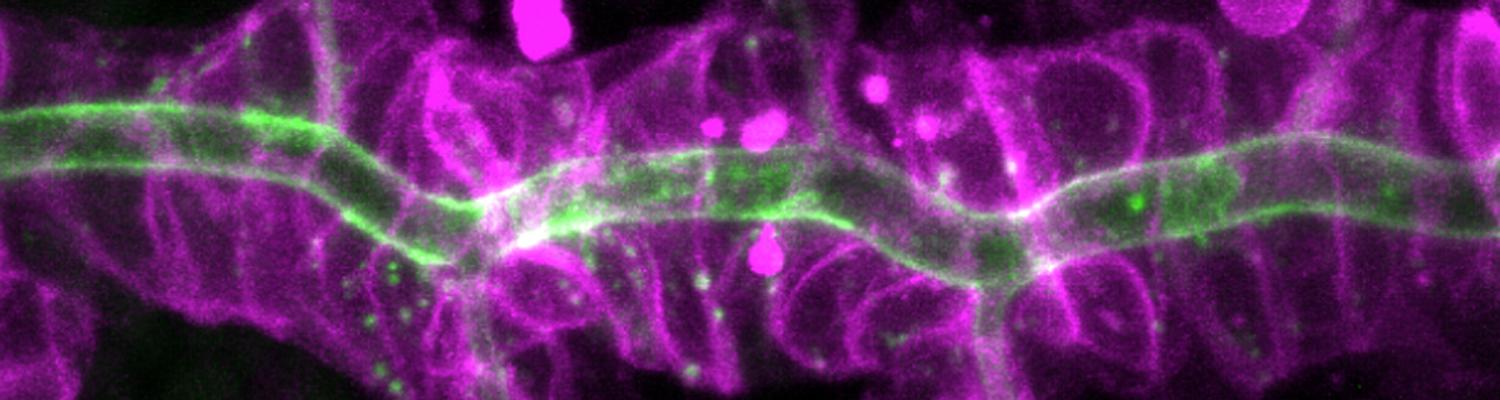
We study how tubular networks develop from initially separate units. In the vascular system separate endothelial sprouts fuse to build tubular bridges (anastomoses). Despite the fundamental role of this process in organogenesis and pathology, the mechanisms that govern epithelial tube fusion are not well understood. Tube fusion events resembling vascular anastomosis formation also occur during development of the tracheal system.
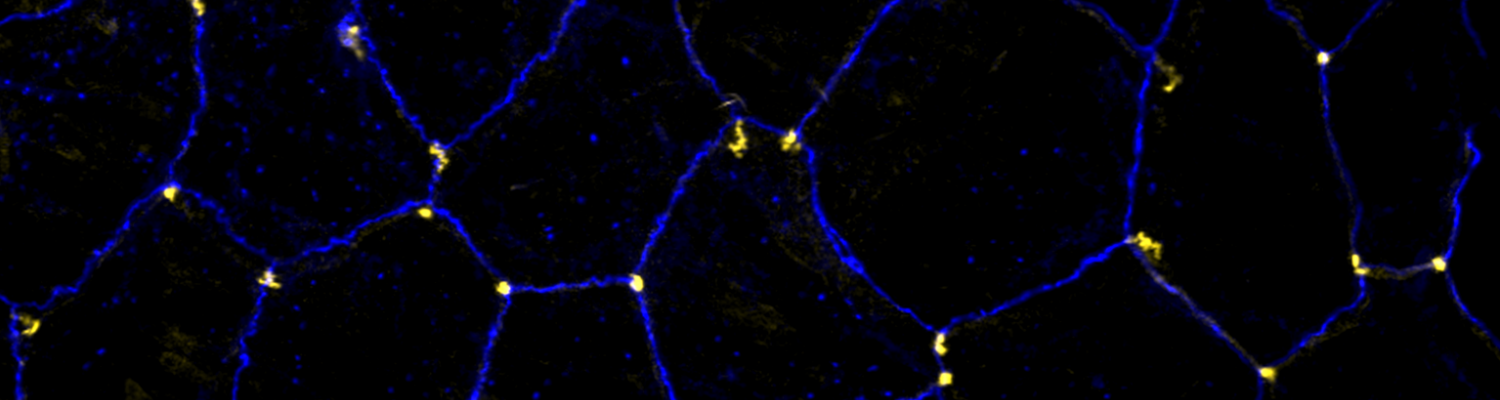
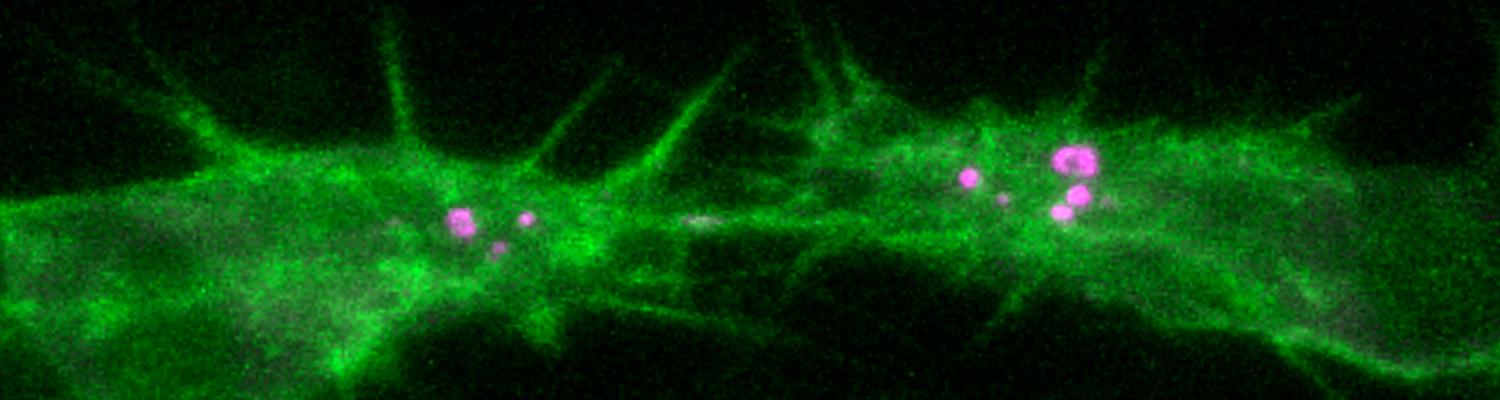
Formation and Function of Epithelial Tricellular Junctions
We investigate the assembly and function of epithelial barriers. Tight junctions in vertebrates and septate junctions in invertebrates form diffusion barriers by sealing the paracellular space between plasma membranes of neighboring cells. Junctions between two adjacent cells (bicellular junctions) have been intensely studied and most of their components are known. However, sealing the epithelium at sites of contact between three cells requires specialized tricellular junctions (TCJs).
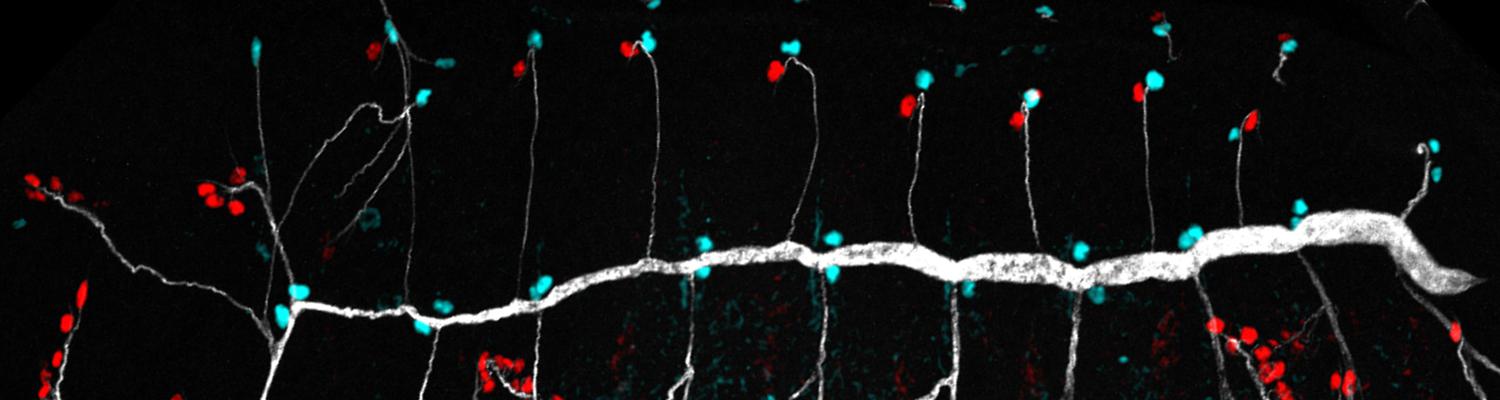
Roles of Hypoxia in Development
Cells are able to sense and respond to oxygen deprivation (hypoxia) by modifying their metabolism to cope with low oxygen levels. The evolutionarily conserved hypoxia response plays a crucial role for survival under changing environmental conditions, as well as in diseases, including cancer, ischemia, and stroke. In Drosophila larvae, hypoxia induces tracheal growth and branching, resembling angiogenic responses induced by hypoxic tumor cells. We use hypoxia-induced tracheal growth as a paradigm to address the interrelation between cellular metabolism and gas exchange.